State of Nevada eCR Program

Purpose
Electronic Case Reporting (eCR) is the automated, real-time generation and transmission of case reports from the electronic health record (EHR) to public health agencies for review and action. eCR improves the detection of outbreaks, allows for earlier interventions while promoting more complete and accurate data transmission. It also diminishes the burden on healthcare providers to report data to local jurisdictions.
eCR enables providers to send infectious disease case reports to the Nevada Division of Public and Behavioral Health (DPBH) per state requirements. On November 9, 2021, DPBH declared its readiness to support data exchange under programs like CMS’s Promoting Interoperability . Nevada uses HL7 electronic initial case report (eICR) standards to meet these requirements and aims to phase out manual reporting over time.

Promoting Interoperability
Promoting Interoperability: In 2011, the Centers for Medicare and Medicaid Services (CMS) established the Medicare and Medicaid Electronic Health Record Incentive Programs to encourage eligible professionals, eligible hospitals, and critical access hospitals to adopt, implement, upgrade, and demonstrate meaningful use of certified electronic health record.
For more information on participant requirements, including ongoing deadlines, visit the Program Requirements page. The Requirements pages outline program specifics for each year.
Nevada Electronic Case Reporting (eCR) Submission Guidelines:
The Nevada Office of State Epidemiology has a registration process for eligible providers and hospitals that intend to submit data for Promoting Interoperability. To register you annual intent to submit electronic case reporting (eCR), please complete the onboarding survey linked below.
Provider Onboarding Registration
You may also choose to email informatics@health.nv.gov with a request to register intent, and you will receive a response with this information.
Important note – this registration encompasses five (5) individual surveys that are available based on what is being reported:
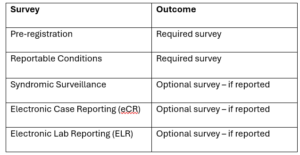
As a helpful tip, the survey includes a “Save and Return Later” feature, allowing you to receive a personalized link via email so you can resume at your convenience.
To maintain compliance, Health Care Organizations must register intent each year. Your reporting facility may be in Pre-Production, Onboarding, Testing and Validation, or Validated Production with eCR to be actively engaged in the eCR process.
Failure to request a letter each year may result in not receiving an Attestation for eCR Compliance letter necessary for the Promoting Interoperability Program for any given year.
How to Participate
Step 1: Ensure that your electronic health record (EHR) system:
- Can produce an electronic initial case report (eICR) that complies with the HL7 3.1 standard for structure and content, and
- Is registered with the Electronic Reporting and Surveillance Distribution system to support triggering and reporting of reportable conditions.
Step 2: Express Your Interest
Take steps to indicate your intention to participate in electronic case reporting (eCR) by:
- Sending an email to the national eCR support team (ecr@cdc.gov)
- Complete the Provider Onboarding Registration. This signifies your notification of intent for Promotion Interoperability with the State of Nevada
Step 3: Set Up the Connection
- Collaborate with your EHR provider to establish a connection to the APHL Integrated Messaging Service (AIMS) hub.
- For assistance, you can contact the national eCR support team via email.
Step 4: Conduct Testing and Validation
Once the connection has been set up, your organization will move into a testing and validation phase. During this phase, your eCR data will be subject to basic testing and validation by AIMS, followed by further testing and validation conducted by the Department of Public and Behavioral Health (DPBH).
Nevada is Currently a Dual Reporting State
- Reporting facilities and partners must continue to report case data through your current means until you are notified by the Nevada Department of Public and Behavioral Health Electronic Case Reporting (eCR) Core Team that it is okay to cease any current reporting practices.
- Additional information on how to report can be found here: Report a Case | Nevada OSE
- Per the state administrative code (Nevada Administrative Code (NAC) Chapter 441A) ALL laboratories and healthcare professionals must report notifiable conditions to the NVDPBH manually and electronically. Manual Reporting can be completed via fax or phone.
- We recommend that your Electronic Health Record (EHR) is turned on for all conditions as Nevada has published all reportable conditions as of January 1, 2025. Please find more information about EHR Triggering here.
- You may email informatics@health.nv.gov for questions or status updates. Thank you in advance for your collaboration.
Resources
The State of Nevada uses Health Level 7 (HL7) Electronic Initial Case Report (eICR) and Reportability Response (RR) standards for electronic case reporting (eCR). Please ensure that your Electronic Health Record (EHR) or Health IT product can support at least one of the HL7 implementation guides shown below:
- HL7 Clinical Document Architecture (CDA) Implementation Guides
- eICR R3.1.1 (HL7 CDA* R2 Implementation Guide: Public Health Case Report – the Electronic Initial Case Report (eICR) Release2, STU Release 3.1.1 – US Realm)
- RR R1.1 (HL7 CDA* R2 Implementation Guide: Reportability Response, release 1, STU Release 1.1 – US Realm)
- HL7 Fast Healthcare Interoperability Resources (FHIR) Implementation Guide
- FHIR eCR R2.1.2 (HL7 FHIR* Implementation Guide: Electronic Case Reporting (eCR), Release 2.1.2 – US Realm)
Other Requirements
- Any senders utilizing the eCR Now FHIR App must update to the latest version at least annually. Release information can be found here: Releases · drajer-health/eCRNow.
- Senders must conduct full triggering of eICRs (i.e., triggering for all conditions) by implementing Electronic Reporting and Surveillance Distribution (eRSD) version 3 or higher.
Important:
- All existing eICR R1.1 senders should update to eICR R3.1.1 by June 1, 2026
- Any new EHR product eCR implementation after January 1, 2026, must use eICR R3.1.1.
Additional Resources:

